Ultrasound energy: a powerful tool for carbohydrate synthesis
Chang and co-workers from Utah State University, USA, have demonstrated that
a large number of conventional reactions commonly employed for the synthesis
and/or functionalization of carbohydrates may be performed under ultrasound
irradiation with remarkable results in terms of yields and reaction time.
(J. Org. 201732-49-2 site Chem. 2006, 71, 5179.
DOI: 10.1021/jo060374w)
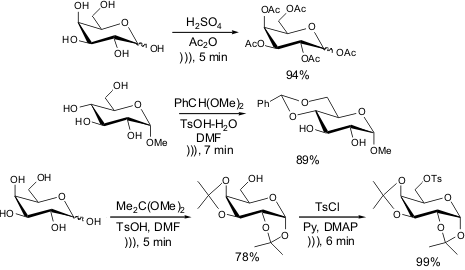
For example, ultrasound-mediated acetylation of hydroxyl groups could be accomplished in
several minutes instead hours required by conventional methods. It is noteworthy
that even the hindered disaccharide trehalose underwent the reaction to afford
the desired acetylated sugar in good yield. Other procedures such as tosylation
and diol protection have worked well by employing such acoustic techniques.
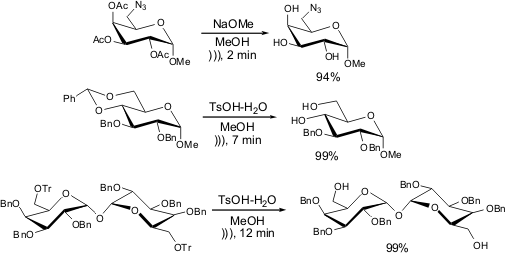
Deprotection reactions can also be carried out
sonochemically. This way, the
cleavage of acetyl and benzylidene groups has been achieved in good yield after
a few minutes. Interestingly, even deprotection of the trityl group could be
achieved smoothly in almost quantitative yield in 12 minutes; whereas the
cleavage of such groups generally demands harsher conditions, it is clear that
ultrasound irradiation helps the reaction proceed to completion.
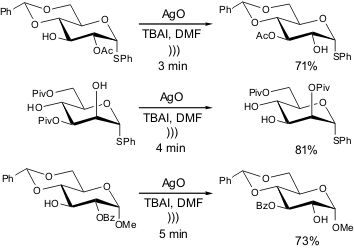
The same group reported earlier that some acyl group migrations within the
carbohydrate scaffold take hours or even days for reaction completion.
(Synlett 2006, 756.
DOI: 10.1055/s-2006-933105)
However, sound-wave activation promotes a
substantial enhancement in the reaction rate; therefore, the procedure afforded
the desired products in several minutes and in good yield under such conditions.
Ultrasound irradiation applied to glucose oligomerization
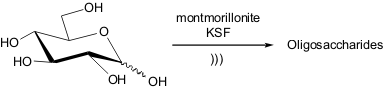
In 2001, the group of Yves Queneau and co-workers discovered a new technique
for glucose oligomerization.
(Ultrason. Sonochem. 2000, 7, 157.
DOI: 10.1016/S1350-4177(99)00035-8)
Firstly, they were interested in developing the
montmorillonite KSF-catalyzed glycosylation reaction of butanol and dodecanol
under ultrasound irradiation. PMID:24187611 1780378-34-8 web Nevertheless, they have found that sound-wave
activation had no significant effect on the glycosylation of butanol, even when
using homogeneous or heterogeneous catalysts. In the case of dodecanol
glycosylation, it was found that reaction under conventional stirring leads to
formation of glycosides preferably, however by employing ultrasound irradiation
the autoglycosylation process took place almost exclusively, and the oligomers
precipitated out from the solution. Furthermore, after a structural analysis it
was found that the oligomers obtained using the acoustic technique were longer
than the ones obtained under silent conditions.
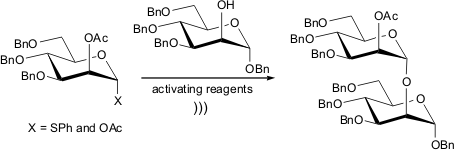
Sonication-assisted oligomannoside synthesis
Christabel T. Tanifum and Cheng-Wei T. Chang from Utah State University, USA,
disclosed a new protocol to get oligomannosides in good yields, short reaction
times and high stereoselectivity.
(J. Org. Chem. ,2009, 74, 634.
DOI: 10.1021/jo8019835)
In the first part of their article they evaluated the
influence of protecting group as well as solvent on the glycosylation of
6-azidohexanol by using a 1-thio-R-D-mannopyranoside as donor. It was found that
in most of cases they observed the selective formation of α-glycosides. In an
attempt to improve the amount of the α-anomer they tested a 4,6-O-benzilidene protected sugar.
Nevertheless, this effort failed and the α-product was formed
exclusively. Based upon this fact, the authors suggest that the sonication
energy is strong enough to overcome the conformational barrier imposed by the
protecting group. Therefore, the use of such an energy source is essential to
keep the α-selectivity of the reaction. Finally, they proceeded with traditional
protection-deprotection protocols and ultrasound-mediated glycosylation
reactions with other carbohydrate molecules to afford trimannosides in good
yields. It is noteworthy that ultrasound-promoted reaction has worked well even
at the last glycosylation step. Moreover, no cleavage of the glycoside bond
during the oligomannoside synthesis was observed.
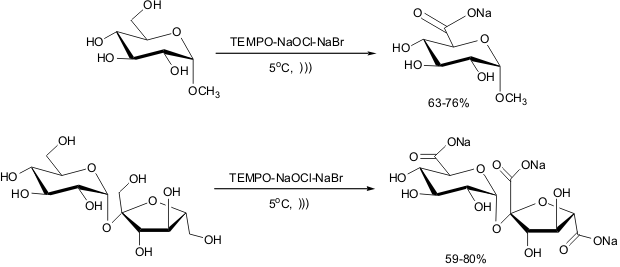
TEMPO-mediated Oxidation of Glucose or Sucrose under Ultrasound Irradiation
The same group cited above has developed a selective chemical oxidation
reaction of the primary hydroxyl group of methyl α-D-glycoside to yield sodium (methyl
α-D-glucopyranoside)uronate by employing sound wave energy.
(J. Mol. Catal. A: Chem. 1999, 150, 31.
DOI: 10.1016/S1381-1169(99)00197-1)
They have obtained 63-76% yields, which depend on the probe diameter. They performed the reaction
from 20 to 500 MHz or 0.008-0.26 (W/mL) acoustic power and around 5°C. They
have used as reactants TEMPO (0.65 mol%), NaBr (0.4 equiv.), NaOCl (2.2 equiv.)
and pH 10.5. Generally, a two-fold rate enhancement was observed in the
sonochemical experiment when compared with the classical conditions. The authors
have also observed that the presence of NaBr is not necessary for the reaction
completion when employing ultrasound irradiation, whereas under silent conditions, such
reagent is quite essential. Therefore, they have tried to rationalize the role
of the sound-waves on this oxidation. As it is well known that
ultrasound-irradiation promotes formation of radical species, they have proposed
this occurs in the formation of the nitrosonium ion from TEMPO; thus enhancing
the reaction rate. Later the same protocol was successfully employed in the
oxidation of sucrose yielding the tricarboxylic acid in good yields.
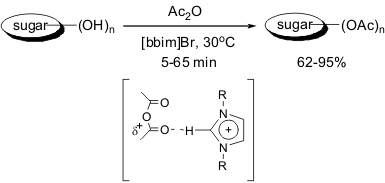
Ultrasound-Ionic Liquid Promoted Acetylation of Sugars at Room Temperature
The acetylation of sugars has been achieved by using ionic liquid at room
temperature in good yields (80-91%). When performed without ultrasound conditions, this
reaction requires longer reaction times (120 min.) for complete conversion. The
reaction proceeds without the addition of acid catalyst because of the acidic
properties of the ionic liquid 1,3-di-n-butylimidazolium bromide ([bbim]Br).
(Green Chem. 2003, 5, 693.
DOI: 10.1039/b308069b)
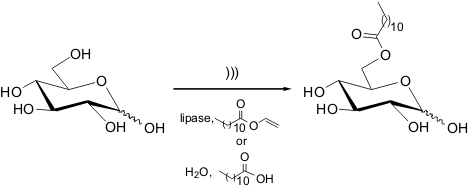
Ultrasound/Lipase/Ionic Liquid-Promoted Synthesis of Sugar Esters
Ha and co-workers reported the enzyme-catalyzed esterification of sugars in
ionic liquids (IL) under ultrasound irradiation.
(Process Biochem. 2008, 43 1009.
DOI: 10.1016/j.procbio.2008.05.001)
In this green method, the lipase catalyzes the transesterification of glucose with vinyl laurate
in the IL [Bmim][TfO] to afford 6-O-lauroyl-D-glucose. In general, the ultrasound-mediated
reactions are 2.6 times faster than the ones performed under conventional conditions. It
is noteworthy that the same reactions could also be done under the same
ultrasound conditions by employing water and lauric acid. Thus, this cheaper protocol might
attract interest from industries that are developing this kind of process.
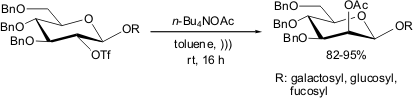
Ultrasound Promoted Displacement of O-Trifyl by O-Acetyl
Fürstner and Konetzki have reported an efficient nucleophilic substitution of
triflate by acetate groups in D-glucopyranose derivatives in the presence of
n-Bu4NOAc and ultrasound energy
(Tetrahedron Lett. 1998, 39, 5721.
DOI: 10.1016/S0040-4039(98)01163-0)
The importance of ultrasound in this reaction has been highlighted;
because no product formation has been observed when the reaction
is performed under silent conditions. Moreover, when the
reaction was carried out for longer times under silent conditions, only
decomposition of the starting materials took place.