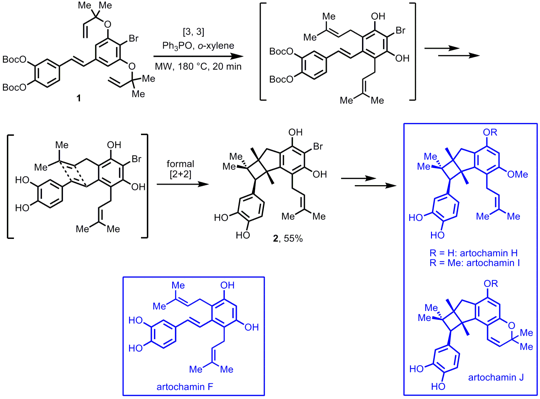
K. C. Nicolaou and his group from the Scripps Research Institute have performed the total synthesis of artochamins F and H-J applying microwave heating for the key step (Angew. Chem. 3-Bromo-5-methylpyrazin-2(1H)-one Chemical name Int. Ed. 2007, 46, 7501. DOI: 10.1002/anie.200702363). DBCO-PEG4-NHS ester Data Sheet After synthesizing stilbene 1, the artochamin skeleton 2 was obtained via a cascade sequence that required catalytic amounts of Ph3PO. The cascade reaction incorporated two consecutiveClaisen rearrangements, collapsing of the Boc groups and a formal [2+2] cycloaddition. Further chemoselective protection of 2 with subsequent methylation afforded artochamins H and I and artochamin J by generation of a benzopyran scaffold. PMID:23715856 If the TBS protected stilbene 1 is employed under the same conditions but without Ph3PO, the cascade reaction stops after the Claisen rearragements, indicating the need of the unprotected hydroxyl groups for the formal [2+2] cycloaddition. After debromination and TBS deprotection, artochamin F was obtained in 92% yield.
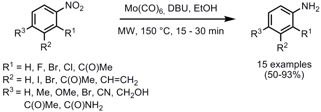
Reduction of Nitro Compounds with Mo(CO)6 and DBU
The group of John Spencer from the University of Greenwich at Medway, UK, has reported on the reduction of nitroarenes to anilines mediated by Mo(CO)6 and DBU (Synlett 2007, 2557. DOI: 10.1055/s-2007-986628). The addition of DBU was essential for obtaining the anilines in high yields due to the facilitated liberation of CO from Mo(CO)6 (33% vs 93% for nitrobenzene). The method proved to be chemoselective and tolerates a broad range of functional groups. By applying microwave heating to 150 °C the reaction time could be reduced from several hours to 15-30 min compared to conventional heating.
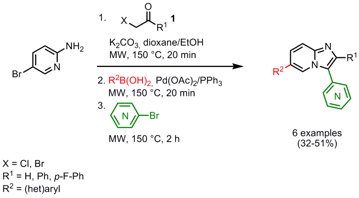
Synthesis of Polysubstituted Imidazo[1,2-α]pyridines
Sabine Berteina-Raboin and coworkers from Université d´Orléans, France, were successful in the synthesis of 2,3,6-trisubstituted imidazo[1,2-α]pyridine derivatives via a one-pot, three-step cyclization/Suzuki coupling/Pd-catalyzed heteroarylation sequence (J. Org. Chem. 2007, 72, 7650. DOI: 10.1021/jo0712603). In previous optimization studies, the best conditions for a one-pot, two-step Suzuki reaction/heteroarylation starting from 6-bromoimidazo[1,2-α]pyridine were evaluated. Pd(OAc)2/PPh3 proved to be superior than Pd(PPh3)4 as catalyst system for the reaction sequence, in addition it is essential to perform the Suzuki coupling as first step. In the three-step sequence, 2-amino-5-bromopyridine was reacted with α-halogenocarbonyls 1 to 6-bromoimidazo[1,2-α]pyridine derivatives followed by the Suzuki coupling and heteroarylation in the same pot, giving the products in somewhat lower yields compared to the two-step sequence.
Synthesis of Carbon-Linked Glycosyl Amino Acids
The groups of Alessandro Dondoni and Alessandro Massi from Università di Ferrara, Italy, have disclosed the synthesis of glycosyl amino acids where the carbohydrate and amino acid residues are linked via a pyridine ring (J. Org. Chem. 2007, 72, 7677. DOI: 10.1021/jo071221r). The synthesis of those C-glycosylmethyl pyridylalanines was accomplished by a Hantzsch-type three-component cyclocondensation of C-glucosyl or C-galactosyl acetaldehydes (1), β-ketoester 2 that incorporates the amino acid residue and enaminoester3. The purification was performed using three different polymer-bound scavengers in one step and one pot. Unreacted enamine was scavenged by sulfonic acid A-15, β-ketoester by the strongly basic anion exchange resin Ambersep and aldehyde by the aminomethylated (AM) resin. Subsequent oxidation employing silica supportedPCC afforded the products in good yields. Attachment of the carbohydrate residue onto the β-ketoester building block and the amino acid residue onto the aldehyde building block, respectively, is also possible.
